RBQM Centralized Monitoring Risk Assessment Risk Management Quality Tolerance Limits Site Management
Made Simple
At TRI we enable Sponsors and CROs alike to unlock the promise of Risk Based Quality Management, reducing manual intervention while improving clinical outcomes.


ABOUT OPRA
RBQM Made Simple, Powerful, and Effective
OPRA from TRI is a RBQM platform that helps organizations to maximize trial efficiency and compliance by clearly identifying critical risks and guiding confident action.
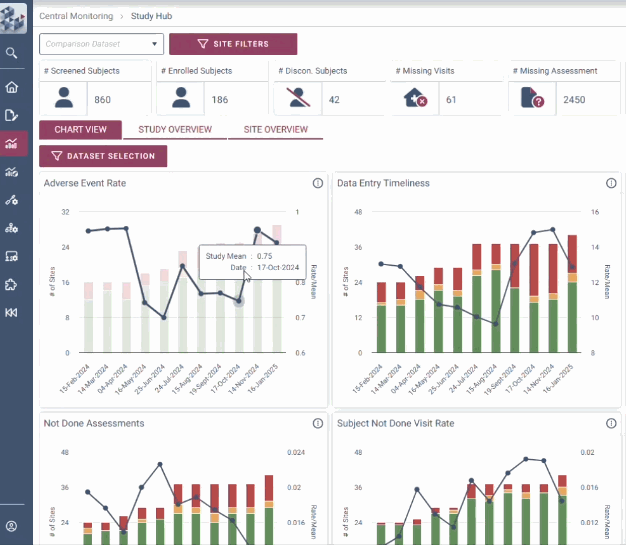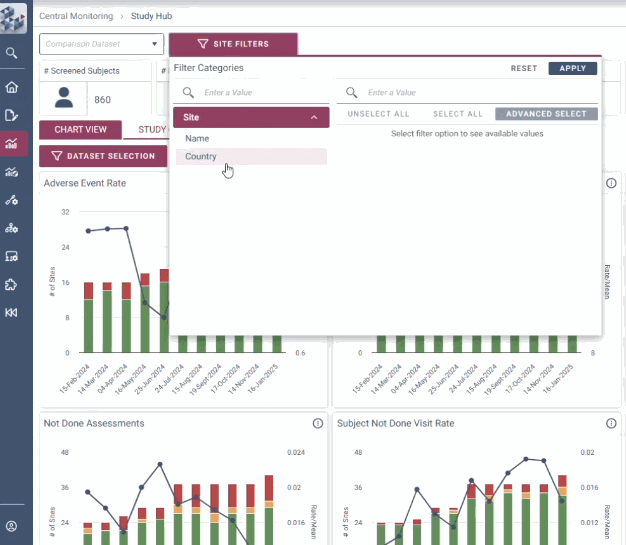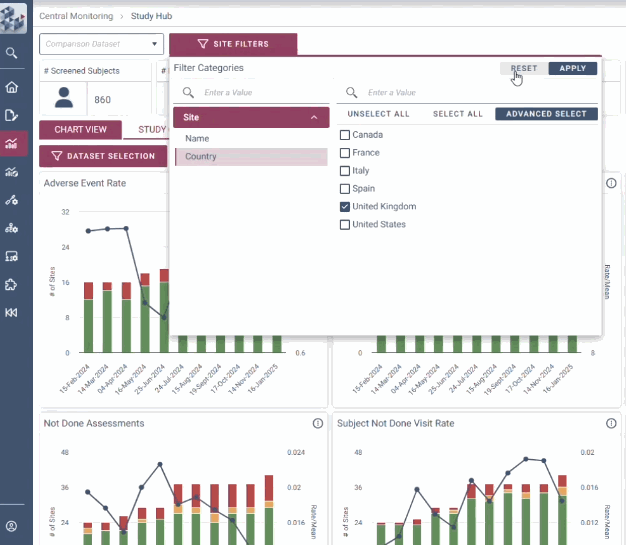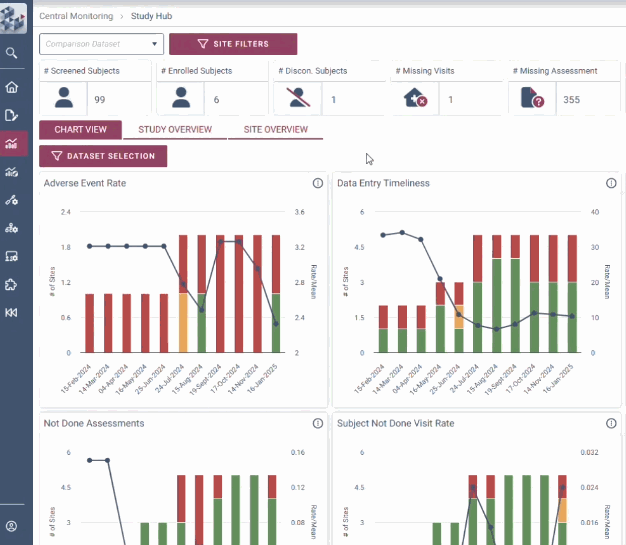
Our RBQM technology delivers crystal clear data visualization, centralized risk management and smart automation, instilling assured decision making for efficiency gains of up to 30%.
With its core modules, OPRA Central Monitoring (OPRA-CM) and OPRA Risk Assessment & Management (OPRA-RAM), teams can seamlessly manage all risks, controls, and actions in one place.
-
Deployed on over 450 clinical trials
-
Secure, Validated, 21 CFR part 11 compliant
-
Designed for Sponsors and CROs running all trial phases
Focus on what matters
TRI helps teams go from overwhelmed to on point. From dynamic data visualizations to a hierarchical database structure, the OPRA platform enables dynamic data analysis from any perspective.
Act with confidence
With TRI teams decisively move from gut-feel to data-driven action. OPRA’s intelligent and dynamic data structure means you can adapt to emerging data and support evidence-based decision making.
Unlock efficiency
Working with TRI teams realise the promise of RBQM – more efficient and safer trials. Together we can cut monitoring costs by up to 30% with OPRA-CM’s streamlined workflows, targeted.
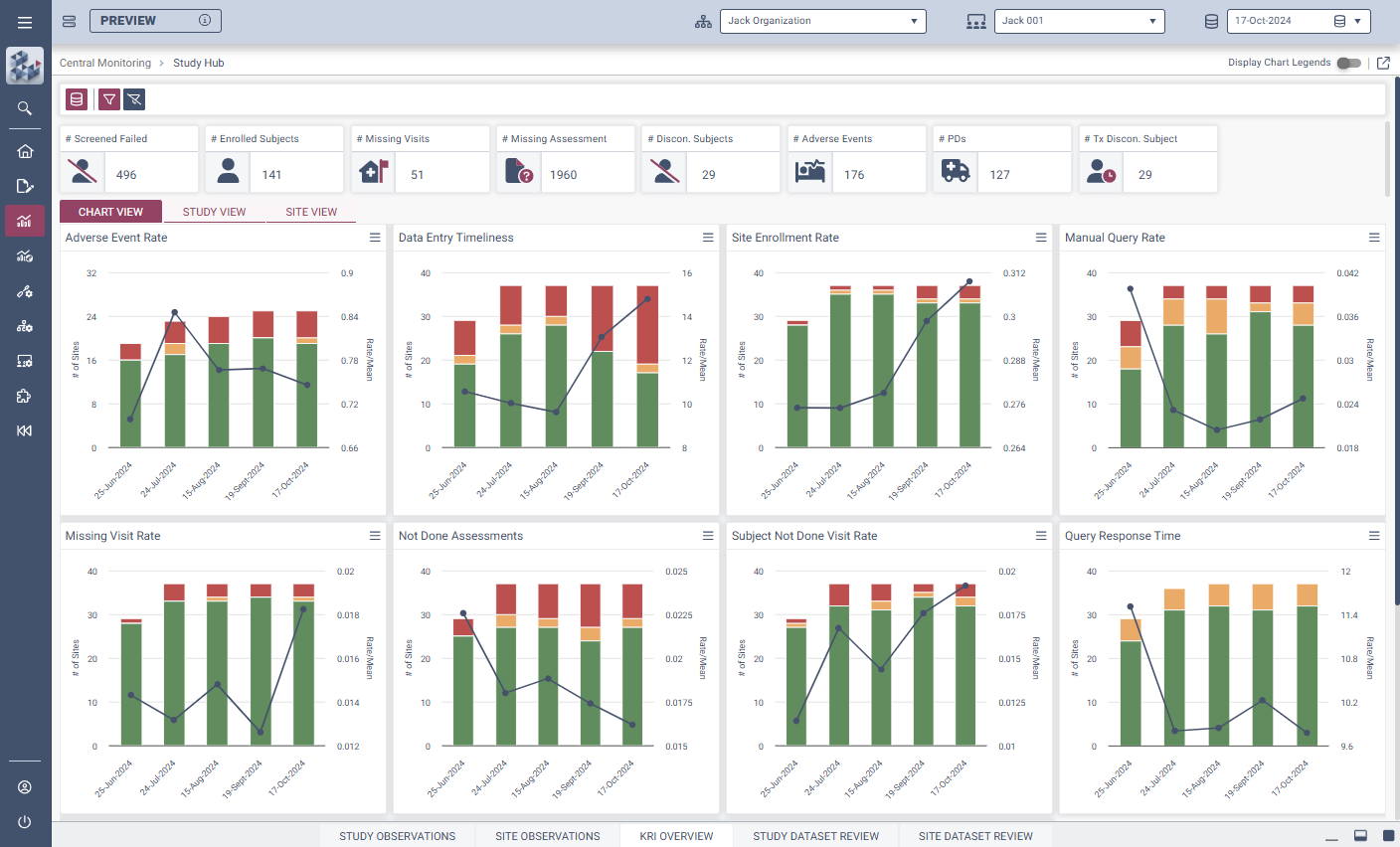
OPRA-CM
Centralized Monitoring, Reimagined
Central Monitoring (OPRA-CM) empowers clinical teams to transform trial oversight with faster, clearer, and more confident decision-making.
Built to drive efficiency and evidence-based action, OPRA-CM helps organizations cut through complexity, focus on critical signals, and stay ahead of risk. Teams gain full control and visibility, regardless of trial size, phase, or geography.
With intuitive data visualizations, real-time trend tracking, and centralized monitoring in one streamlined interface, OPRA-CM eliminates the noise of traditional methods and puts the spotlight on what matters most.
-
Enable instant focus into what matters most
-
Make data-driven decisions with confidence
-
Achieve efficiency gains of up to 30%.
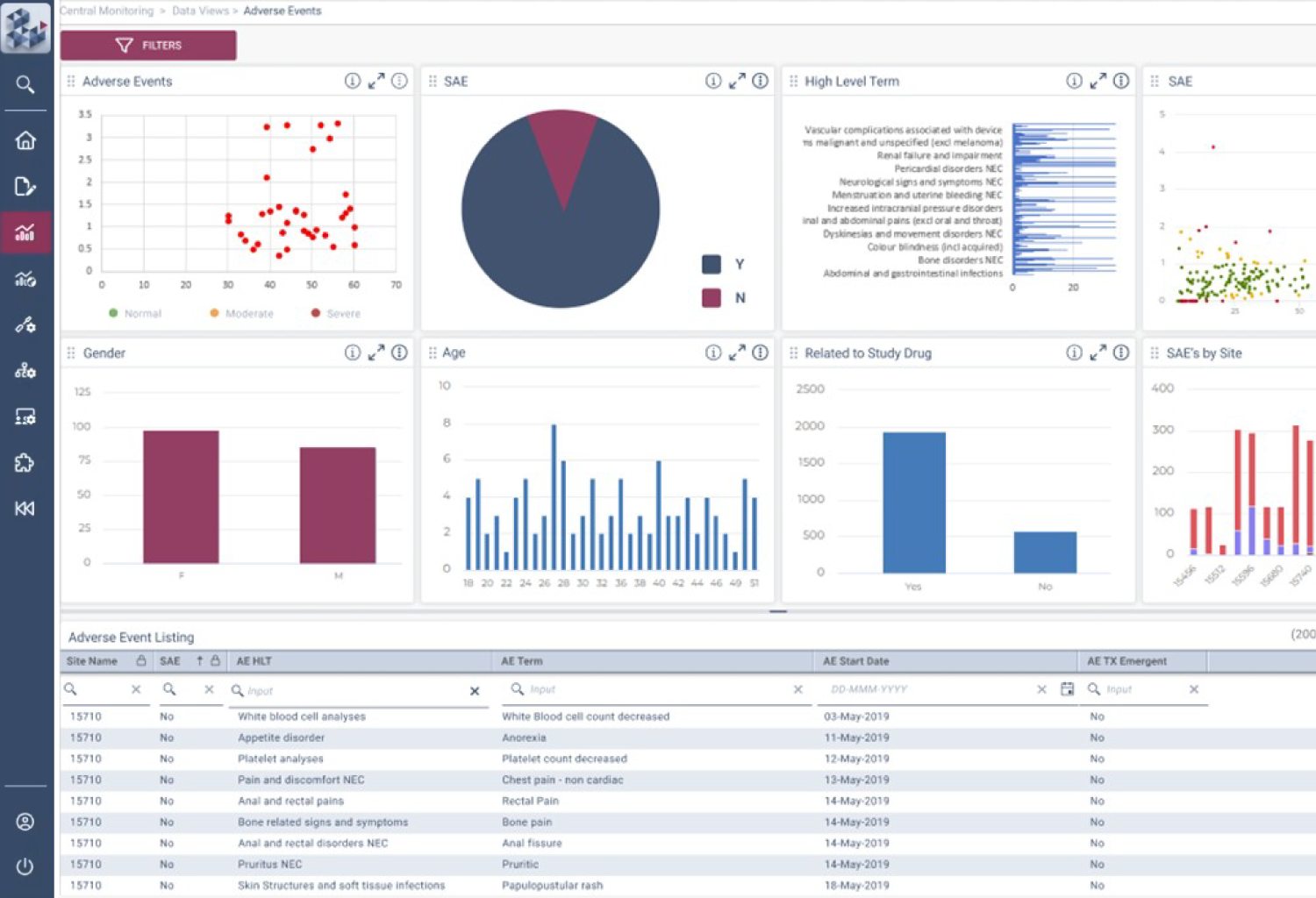
OPRA-RAM
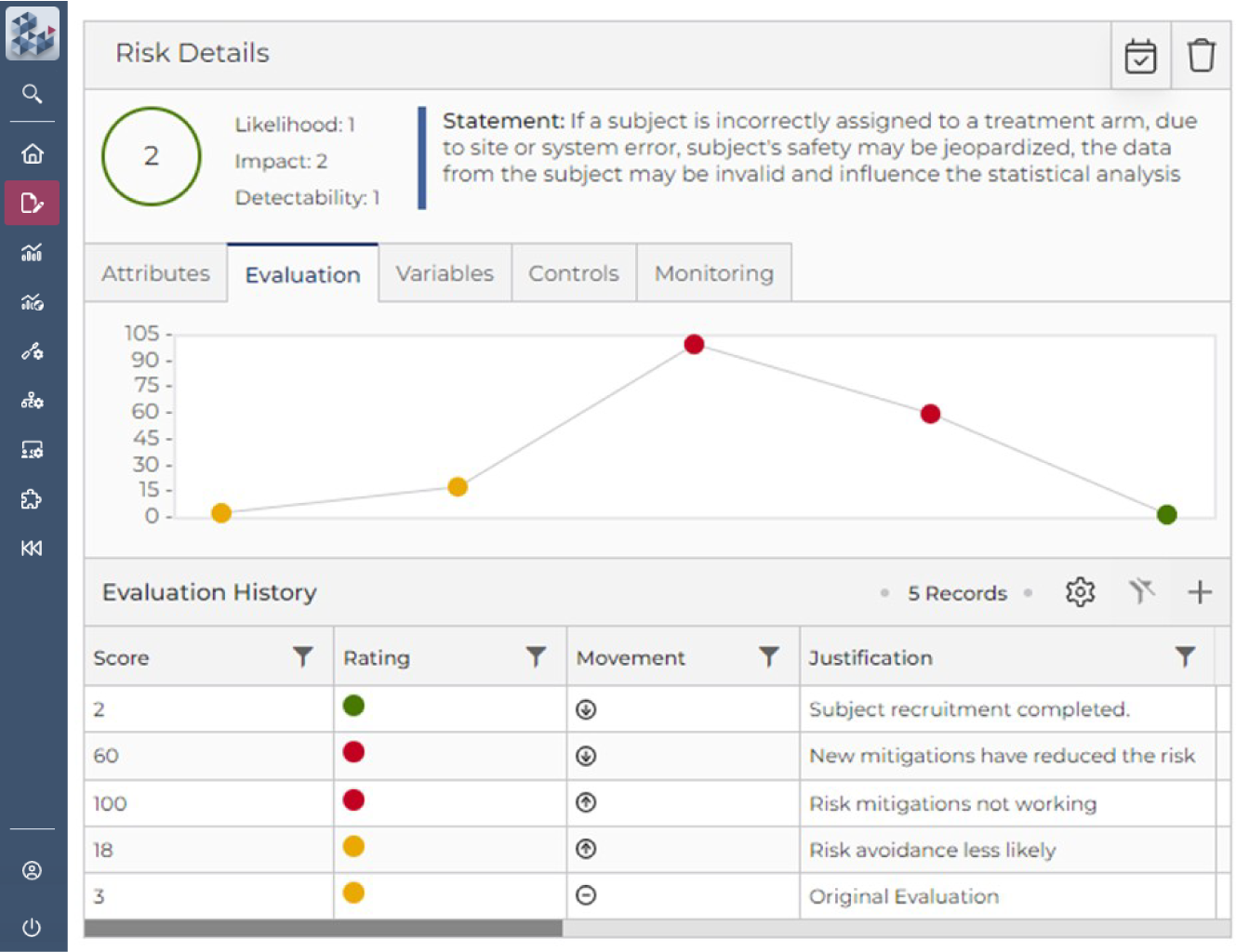
Risk Assessment & Management Simplified
OPRA Risk Assessment & Management (OPRA-RAM) simplifies and strengthens trial management by pulling together all risks, controls and actions into one easy-to-use platform.
OPRA-RAM helps teams work more efficiently, stay compliant, and focus resources where they’re needed most. With centralized activity management, the platform enables clear risk-variable relationships, tracks trends over time, and assesses control effectiveness to support confident, proactive decision-making.
Designed for trials of any scale or phase, OPRA-RAM ensures consistency, speed, and full-cycle visibility across risk processes.
-
Quickly prioritize risk and implement controls
-
Focus resource on critical risks and activities
-
Demonstrate proactive risk management
SUBJECT MONITORING
Subject Monitoring Precision in RBQM
In early phase trials, the ability to oversee the safety and progress of each patient is paramount. OPRA-SM is designed to idenitify and manage patient risk, where traditional site-based Central Monitoring approaches don't work. Medical monitoring of patient data can be inefficient and error-prone, but those problems go away with OPRA-SM's unique views on data changes and patient journeys.
-
Enhanced patient safety
-
Improved data integrity
-
Greater operational efficiency
Compare OPRA
to traditional monitoring solutions
Traditional Monitoring
OPRA
Proactive
Proactively identifies and addresses risks throughout the trial, preventing issues before they occur.
Comprehensive
Takes a holistic approach, considering risks across the entire trial, from protocol design to data analysis.
Data-Driven
Relies on data and analytics to make informed decisions about risk mitigation.
Flexible
Adapts to changes and unforeseen circumstances, maintaining quality in trials.
Adaptable
Allows for tailored risk assessments and mitigation strategies based on trial-specific factors.
Transparent
Promotes transparency by documenting risk assessments, mitigation plans, and decision-making processes.
Frequently asked questions
We've got the answer
Explore our FAQs for answers and expert advice on RBQM.
TESTIMONIALS
RBQM Success Stories
Hear directly from our partners about how we've helped them.


What truly set TriTrials apart from other vendors was their unparalleled commitment to a quality deliverable helping us effectively apply a novel approach to our operations. They achieved a deep understanding of our objectives by partnering with us on a rigorous change management process which culminated in a shared vision for the integration of their risk assessment and central monitoring solution.

The Worldwide Clinical Trials team is pleased with our continued partnership with TRI and we are excited about the milestone of having 150 clinical trials in TRI's Risk-Based Quality Management platform. This is a key benefit for our customers as it improves data quality, trial efficiency, and patient safety.

RBQM NEWS
Clinical trial quality news and views
Subscribe to get the latest RBQM insights sent to your mailbox.
RBQM is redefining how clinical trials are designed, enabling smarter, more resilient research.
Explore how sponsors and CROs must rethink collaboration and embed quality into every layer of trial execution.
Explore how E6 (R3) reshapes trial design, oversight, and culture, and why the industry must be ready to respond.




















